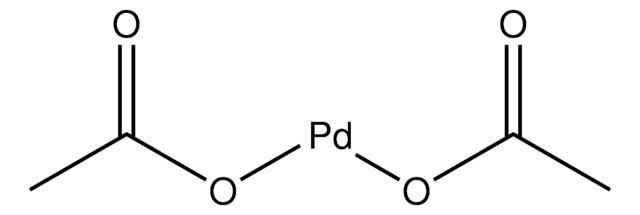
Modular synthesis of simple cycloruthenated complexes with state-of-the-art performance in p-type DSCs - Journal of Materials Chemistry C (RSC Publishing) DOI:10.1039/C6TC03874C

The surprisingly facile formation of Pd( i )–phosphido complexes from ortho -biphenylphosphines and palladium acetate - Dalton Transactions (RSC Publishing) DOI:10.1039/C8DT04926B
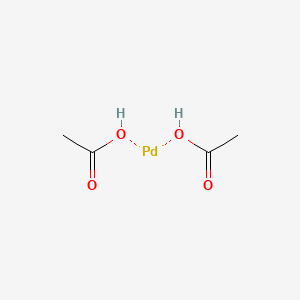
![Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals](https://www.strem.com/uploads/web_structures/46-2033.gif)
Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals

Pd-catalyzed Suzuki–Miyaura reactions of aryl halides using bulky biarylmonophosphine ligands | Nature Protocols

Decarbonylative Fluoroalkylation at Palladium(II): From Fundamental Organometallic Studies to Catalysis | Journal of the American Chemical Society
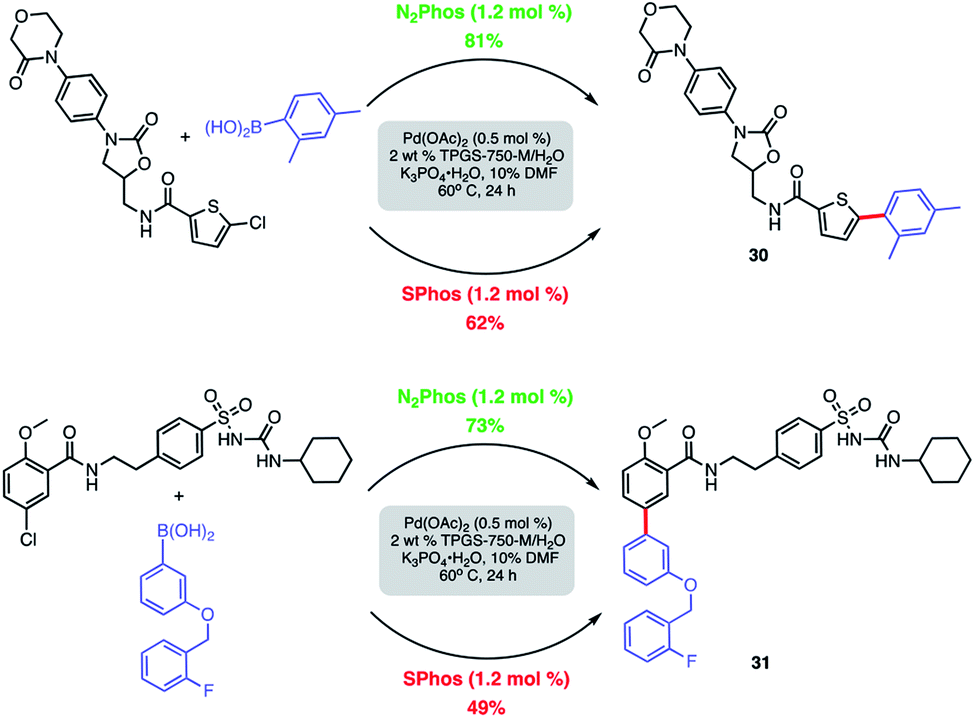
N 2 Phos – an easily made, highly effective ligand designed for ppm level Pd-catalyzed Suzuki–Miyaura cross couplings in water - Chemical Science (RSC Publishing) DOI:10.1039/D0SC00968G
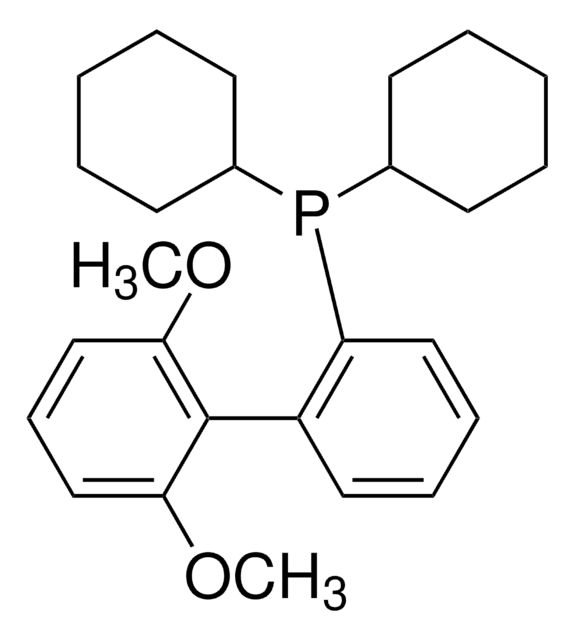
![Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals](https://www.strem.com/uploads/web_structures/46-2040.gif)
Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals

Suzuki–Miyaura Cross‐Coupling under Solvent‐Free Conditions - Asachenko - 2013 - Advanced Synthesis & Catalysis - Wiley Online Library

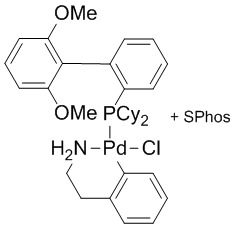
Development of Palladium Precatalysts that Efficiently Generate LPd(0) Active Species - Shaughnessy - 2020 - Israel Journal of Chemistry - Wiley Online Library
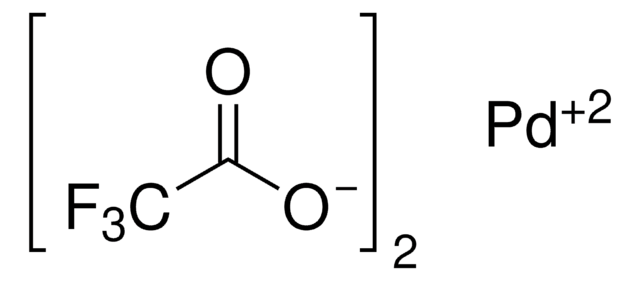
![Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals Palladium(II) acetate/2-dicyclohexylphosphino-2,6-dimethoxy-1,1'-biphenyl ( SPhos)/potassium phosphate admixture [CatKit single-use vials - 1.96 wt% Pd(OAc)2] | Strem Chemicals](https://www.strem.com/uploads/web_structures/46-2030.gif)